Main Research Questions
- How do patient-derived cellular models of brain disease differ from those of healthy individuals?
- How can patient-derived cellular models of brain disease be applied to identify novel treatments?
- How can large-scale electronic health records be used efficiently to identify novel treatments, risk genes, or predictors of treatment response?
Research Approaches
We are developing methods for modeling microglia-mediated synaptic pruning in vitro, and are using them to understand pruning abnormalities in schizophrenia and to identify novel treatments.
- We developed one of the largest cellular biobanks in the world for the study of brain diseases, a key resource for understanding how disease-associated genes contribute to risk, as well as screening for new treatments.
- We are now using our scalable human synaptic pruning in vitro model to investigate the potential effects of genes implicated by postmortem and genomic studies of schizophrenia risk genes
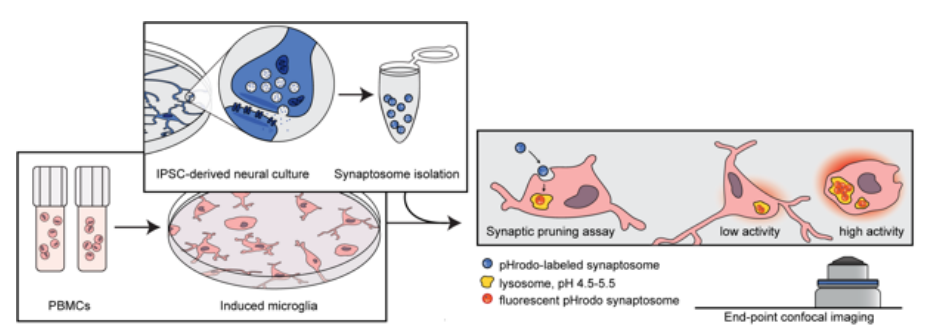
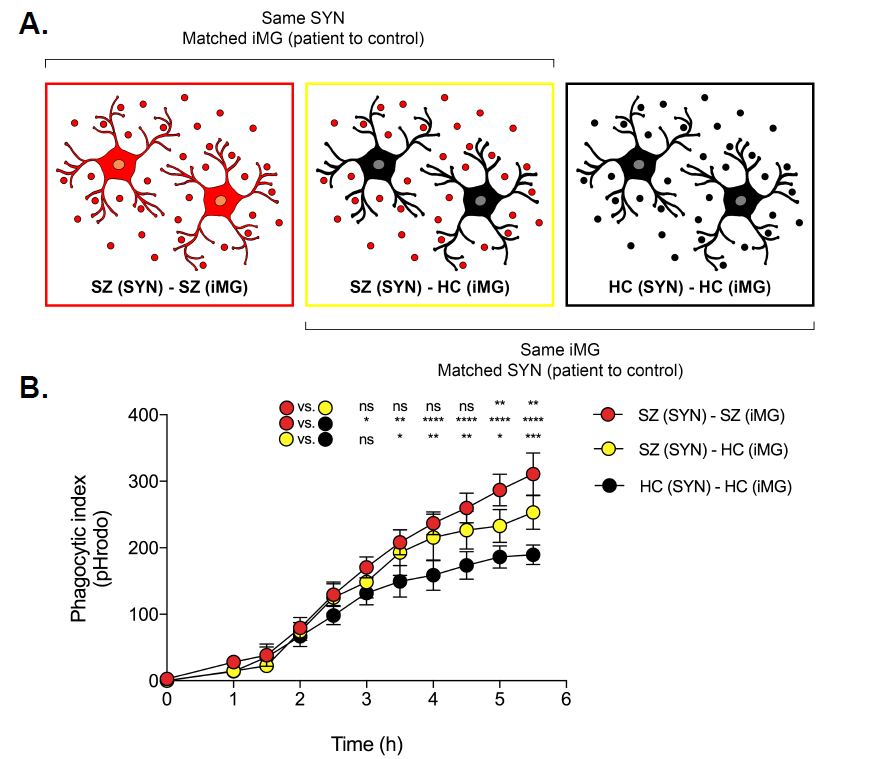
We are developing new tools for visualizing risk in clinical populations, allowing clinicians to make better use of emerging clinical and genomic predictors.
We are developing ways to make better use of health records and biobanks to drive genetic discovery – for example, understanding the effects of antidepressants on cardiac rhythms.
- We helped lead the identification of the first risk genes for major depressive disorder
- We developed machine learning approaches that apply electronic health records to predict suicide and other causes of mortality in individuals with psychiatric illness.
